|
3/31/2024 0 Comments Density of water gl 
Measurements are immensely time-consuming and might take one to two days. This “instrument” is very expensive and not suitable for industry. For example, if you had a metal cube with mass 7.0 g and volume 5. To solve for density, simply follow the equation 1.5.1. However, a hydrostatic balance is a huge, complex apparatus with a concrete foundation that needs to be insulated and extensively temperature-controlled. Grams per centimeter cubed is equivalent to grams per milliliter (g/cm3 g/mL). The density of water varies according to temperature and the degree of purity. Hydrostatic balance: Hydrostatic balances are used in special facilities, for example the National Bureau of Standards, because they are incredibly precise and reliable. Density of pure water is a constant at a certain temperature not depending on sample. As is the case with pycnometers, a large sample amount is needed and the hydrometer can break easily. However, the scale can easily be misread and a temperature correction is necessary. 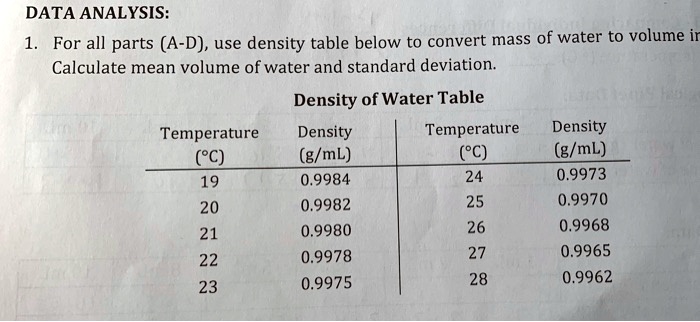
Hydrometer: Hydrometers are affordable and provide a very fast concentration measurement. All in all this method is quite time-consuming and it is only possible to determine the apparent density of the sample, so the buoyancy of air has to be compensated. Another drawback is the temperature control of the sample of which a large amount (up to 100 mL) is needed. However, to achieve highly accurate results a very skilled operator is needed as well as a very precise balance. Pycnometer: A pycnometer is an affordable measuring device that gives highly accurate measurement results. These points make this method very time-consuming and not user-friendly. Another drawback of titration is the use of aggressive chemicals and the need of costly consumables. Further, a special setup depending on each individual sample is needed. A limited concentration range is covered, so the sample has to be diluted accordingly. 1 kilogram/cubic meter is equal to 0.001 g/mL, or 1 g/L. We assume you are converting between gram/millilitre and gram/litre.You can view more details on each measurement unit: g/mL or g/L The SI derived unit for density is the kilogram/cubic meter. How many g/mL in 1 g/L The answer is 0.001. Titration: A common disadvantage of titration is that it requires a skilled operator with chemical know-how. More information from the unit converter. This refers to the extract content in the product, typically beer wort, and is more or less the same. This means that there are 700 mL of ethanol in 1 L solution.Ī common concentration unit in the brewing industry for describing the concentration of beer and wort is °Plato or ☋alling. In the pharmaceutical industry, for example, a disinfection solution may contain 70 % ethanol ( V/ V). This, of course, is a temperature-dependent unit. If you look at a bottle of beer, you will typically see the alcohol content reported in %V/V. Divide the mass by the volume, and you will have the density of water. While most industries use % weight/ weight, the alcohol industry mainly reports their concentration in % volume/ volume. Temperature can be ignored here, since temperature does not have any influence on weight or mass. One example: 5 % sulfuric acid means that there is 5 gram of sulfuric acid in 100 gram of the solution. Generally this means: gram of the component per 100 gram of the solution. Density of water as a function of temperature. In the chemical field concentrations are often reported in % weight/ weight or % mass/ mass. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
